Customer:
Nobel Biocare
Smart Labels Boost Nobel Biocare
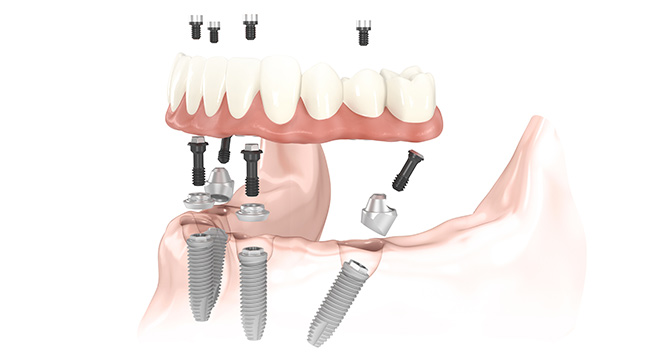
Nobel Biocare makes a wide range of titanium screws used to fix tooth implants into people’s mouths. It is no coincidence that the medicine-technological industry is stringently supervised. In order to sell medicine-technological equipment, approvals from governmental agencies in the target countries are required.
"Version handling and traceability, covering all changes at both label data and label layout levels are some of the advantages of Prevas’ system Snitcher Medical", says Jonas Edlund, System Manager for Snitcher Medical at Nobel Biocare.
Assignment: A standard GMP compliant labeling system.
An important aidto approval is certifications in quality, and the environment from international organizations. Keeping tabs on your development and manufacturing processes then becomes essential. Because when you do get your product approved in a country, you must subsequently be able to show that all the changes you make hence are implemented correctly, or else you will automatically loose your approval, the moment you improve your product. An important component of any product is its markings. For this reason, an appropriate marking system, complying with the GxP rules governing the pharmaceutical and medical-equipment industries, is essential.
“Version handling and traceability, covering all changes at both label data and label layout levels are some of the advantages of Prevas’ system Snitcher Medical,” says Jonas Edlund, System Manager for Snitcher Medical at Nobel Biocare.
History of all labels, printed, by whom and when, is another essential function, as is support of review, approval and activation of label versions. Nobel Biocare has bought a group-wide license from Snitcher Medical, covering Nobel Biocare’s global production sites, its research and development location and its external suppliers. The server-based architecture of the system facilitates the groups’ global label handling due to the fact that Nobel Biocare is able to design all labels centrally and then provide its internal production sites with the activated labels they need and control which labels external users should be able to print.
“External suppliers may be located in the USA or in Europe. The Internet is used for communications and all transactions are protected by VPN tunneling,” says Edlund.
By virtue of the fact that the new system is integrated with Microsoft’s Active Directory, the administration of users is facilitated. In addition, integration with Nobel Biocare’s administrative system ensures safe orders-based labeling. Variable batch data, e.g. Batch No. and Expiry date are added to the labels automatically. Another advantage is that the new version of Snitcher Medical is able to manage 2D, DataMatrix barcodes. These may contain details of product type, country of origin, batch No., date of manufacture and use-before date.
“The DataMatrix codes offer many advantages when it comes to cramming a lot of information into a small area and using these in your labeling paves the way to satisfying future requirements for traceability,” says Ulrika Magnusson at Prevas.
Jonas Edlund of Nobel Biocare agrees:
“Marking our products with DataMatrix’ codes and complying with the GS1 standard are important services to our customers; it gives us an edge,” he adds.
Edlund says that he is happy with Prevas as supplier:
“Prevas has been responsive to our requirements and we have cooperated fruitfully for quite a time, cooperation that has yielded both stability and quality. It is important to us that, when we do get our products approved, we maintain those approvals over time, despite the fact that we change the system continuously. Snitcher Medical helps us do just that.”
Do you want to know more about this project or find out what Prevas can do for you, please contact us.
CONTACT US
Peter K Johansson, e-mail


Related cases
-

Kenvue
Johnson & Johnson’s Consumer Business Spin-Off to Kenvue – Prevas Leads the IT Application Migration for Kenvue's Helsingborg Factory
-

Hydroscand
Flexible production system for Hydroscand
-

Meritor
eLIPS gives Meritor full control over advanced production logistics
-

GKN Automotive
Flexible internal logistics solution celebrates 20 years of faithful service


